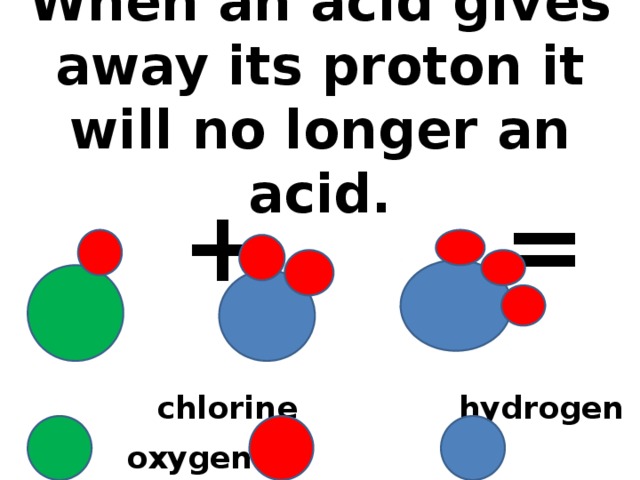
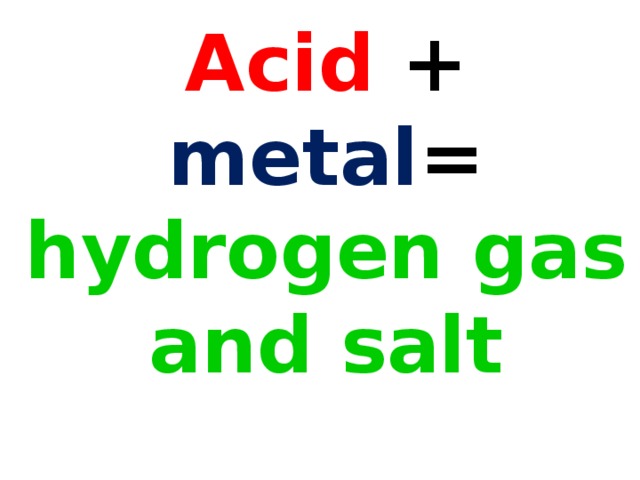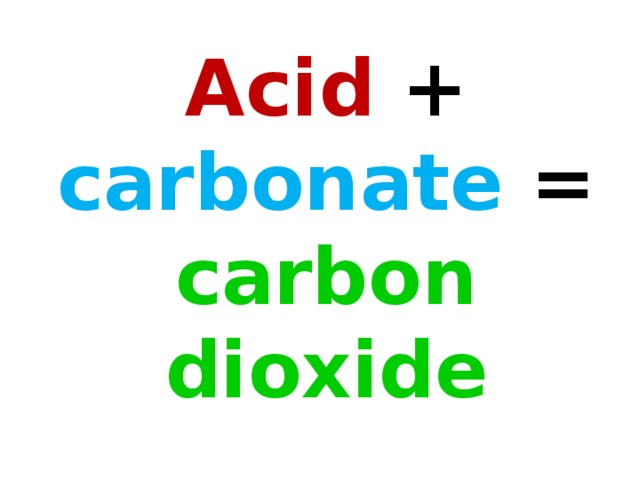Обучение основным понятиям кислоты и их взаимодействие с другими веществами.
Создайте Ваш сайт учителя Видеоуроки Олимпиады Вебинары для учителей
"Понятие кислот и их взаимодействие с другими веществами"
Вы уже знаете о суперспособностях современного учителя?
Тратить минимум сил на подготовку и проведение уроков.
Быстро и объективно проверять знания учащихся.
Сделать изучение нового материала максимально понятным.
Избавить себя от подбора заданий и их проверки после уроков.
Наладить дисциплину на своих уроках.
Получить возможность работать творчески.
Просмотр содержимого документа
«Acids»
Просмотр содержимого презентации
«acids»
Полезное для учителя
Распродажа видеоуроков!
1850 руб.
3080 руб.
1660 руб.
2770 руб.
1610 руб.
2690 руб.
1850 руб.
3080 руб.
ПОЛУЧИТЕ СВИДЕТЕЛЬСТВО МГНОВЕННО
* Свидетельство о публикации выдается БЕСПЛАТНО, СРАЗУ же после добавления Вами Вашей работы на сайт
Удобный поиск материалов для учителей
Проверка свидетельства