Презентация включает элементы английского языка при обучении физики. Презентация содержит задания с приемами технологии критического мышления
Создайте Ваш сайт учителя Видеоуроки Олимпиады Вебинары для учителей
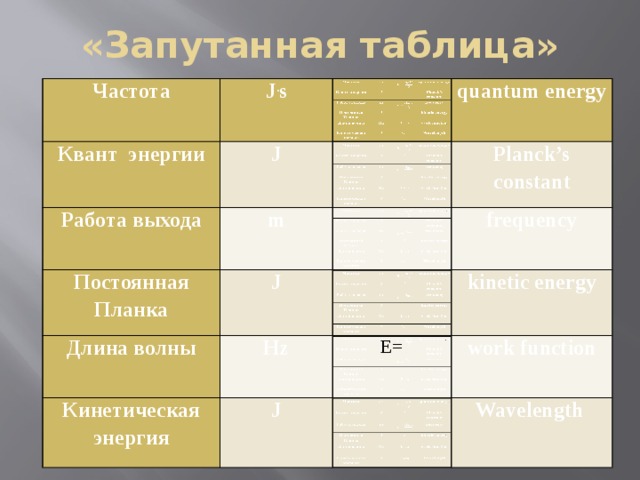
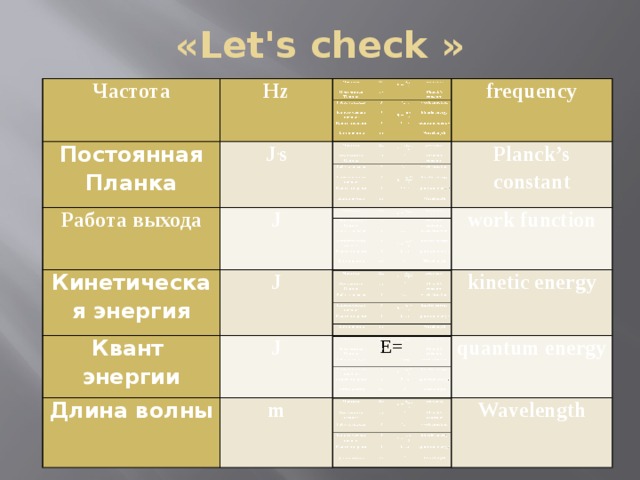
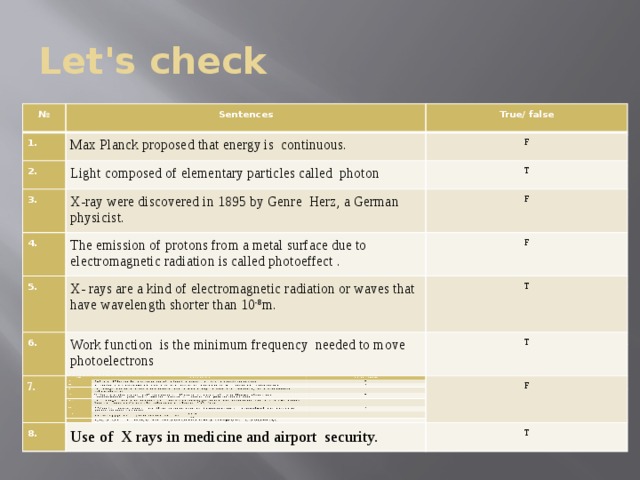
Презентация урока-обобщение 9 класс «Световые кванты. Фотоэффект»
Вы уже знаете о суперспособностях современного учителя?
Тратить минимум сил на подготовку и проведение уроков.
Быстро и объективно проверять знания учащихся.
Сделать изучение нового материала максимально понятным.
Избавить себя от подбора заданий и их проверки после уроков.
Наладить дисциплину на своих уроках.
Получить возможность работать творчески.
Просмотр содержимого документа
«Презентация урока-обобщение 9 класс «Световые кванты. Фотоэффект»»
Полезное для учителя
Распродажа видеоуроков!
1670 руб.
2780 руб.
1610 руб.
2690 руб.
1520 руб.
2530 руб.
1670 руб.
2780 руб.
ПОЛУЧИТЕ СВИДЕТЕЛЬСТВО МГНОВЕННО
* Свидетельство о публикации выдается БЕСПЛАТНО, СРАЗУ же после добавления Вами Вашей работы на сайт
Удобный поиск материалов для учителей
Проверка свидетельства